FDA Approves Regeneron's High-Dose Aflibercept, Eylea HD
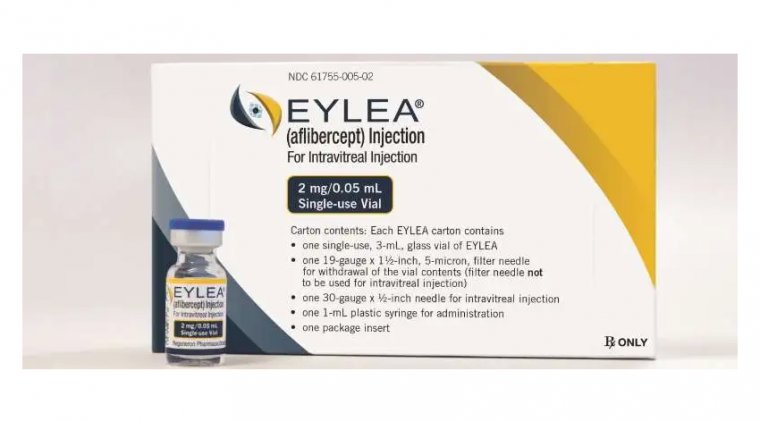
Regeneron has received FDA approval for Eylea HD (aflibercept 8 mg) in a significant development anticipated to substantially extend dosing intervals for patients with retinal diseases such as wet age-related macular degeneration (AMD), diabetic macular edema (DME), and diabetic retinopathy (DR).
The recommended dosing regimen for Eylea HD is 8 mg (equivalent to 0.07 mL of a solution containing 114.3 mg/mL) every 4 weeks during the initial 3 months for all indications. Subsequently, for wet AMD and DME, the dosing frequency is adjusted to 8 mg every 8 to 16 weeks, while for DR, it is recommended to administer 8 mg every 8 to 12 weeks.
“The FDA approval of Eylea HD is an important advancement in retinal care,” Peter Kaiser, MD, Chaney Family Endowed Chair in Ophthalmology Research at the Cole Eye Institute and Professor of Ophthalmology at Cleveland Clinic Lerner College of Medicine, said in a company news release. “With Eylea HD, patients with wet age-related macular degeneration or diabetic retinal disease can now receive less frequent injections after their initial monthly doses and still experience the similar visual gains, anatomic improvements and safety profile of Eylea.”
Eylea HD is set to be priced at a wholesale acquisition cost (WAC) of $2,625.00 for each single-use vial. Regeneron has communicated that the projected annual list price for Eylea HD, when used according to its indications, is expected to fall "within the range of or below" that of the regular Eylea. The annual expenses will differ based on various elements, encompassing the dosing regimen prescribed for individual patients. Regeneron has expressed its dedication to facilitating access to Eylea HD for patients who require the medication.
Approximately a month following the issuance of a complete response letter (CRL) by the FDA to Regeneron, wherein approval for aflibercept 8 mg was withheld, citing the need for further examination of inspection findings at a third-party filler, the approval has now been granted. The CRL did not pinpoint any concerns pertaining to the clinical effectiveness or safety of aflibercept 8 mg, trial design, labeling, or drug substance manufacturing.
No additional clinical trials or data were requested. Regeneron clarified that the CRL was issued solely due to unresolved observations stemming from an FDA inspection at a third-party contract manufacturing entity, Catalent, which Regeneron had engaged to carry out vial-filling for aflibercept 8 mg.
About the EYLEA HD Clinical Trial Program
The FDA's approval of Eylea HD is based on the 48-week findings from two pivotal trials known as PULSAR and PHOTON. These trials were double-masked and active-controlled, assessing Eylea HD in comparison to Eylea (aflibercept) injection 2 mg. Both the PULSAR study involving wet AMD (N=1,009) and the PHOTON study involving DME (N=658) successfully achieved their primary objectives. E
ylea HD, when administered with dosing intervals of both 12 and 16 weeks after three initial monthly doses, demonstrated vision gains that were noninferior and clinically equivalent to those achieved with an Eylea 8-week dosing regimen after the initial monthly doses (3 doses in PULSAR and 5 doses in PHOTON). The majority of patients initially assigned to Eylea HD with 12- or 16-week dosing intervals (following three initial monthly doses) were able to uphold these dosing schedules up to the 48-week mark.
Frequently observed adverse reactions (≥3%) in patients who received Eylea HD included cataract, conjunctival hemorrhage, increased intraocular pressure, discomfort/pain/irritation in the eye, blurred vision, vitreous floaters, vitreous detachment, corneal epithelium defect, and retinal hemorrhage.
“More than 10 years ago, we fundamentally changed the way that certain serious retinal diseases were treated with the introduction of Eylea. With the approval of Eylea HD, we have elevated the high standard that Eylea set,” George D. Yancopoulos, MD, PhD, Board co-Chair, President and Chief Scientific Officer at Regeneron, and a principal inventor of Eylea, said in the news release. “Our continued commitment to retinal diseases resulted in an important scientific innovation – evolving the proven efficacy and safety of Eylea into a new treatment, Eylea HD, that provides lasting vision control with even fewer injections to further benefit those living with wet age-related macular degeneration or diabetic retinal diseases."
Eylea HD is jointly developed by Regeneron and Bayer AG. Within the United States, Regeneron holds exclusive rights to both Eylea and Eylea HD. Outside of the U.S., Bayer possesses the exclusive marketing rights, leading to a shared profit distribution between the two companies for sales of Eylea and Eylea HD after securing regulatory endorsements.
Currently, aflibercept 8 mg is undergoing regulatory evaluation in Europe and Japan. Furthermore, the companies plan to submit applications to additional regulatory bodies in various countries.
*Stay in the loop and make sure not to miss real-time breaking news about ophthalmology. Join our community by subscribing to OBN newsletter now.
(1).jpg)