Researchers Discover a Nanobody to Treat Retinitis Pigmentosa
A group of researchers from the University of California, Irvine, has made a significant discovery: an exceptional antibody that could pave the way for treating Retinitis Pigmentosa. This condition results in the deterioration of central vision, as well as night and color vision.
Published Study on Rhodopsin and Nanobody Binding
The scientific paper titled "Structural basis for the allosteric modulation of rhodopsin by nanobody binding to its extracellular domain" has been published in the journal Nature Communications. Notable authors of the research include Arum Wu, PhD, David Salom, PhD, John D. Hong, Aleksander Tworak, PhD, Philip D. Kiser, PharmD, PhD, and Krzysztof Palczewski, PhD. This work was carried out within the Department of Ophthalmology at the Gavin Herbert Eye Institute of the University of California, Irvine. The study also saw collaboration with Jay Steyaert, PhD, from the Vrije Universiteit Brussel (VUB).
What is Retinitis Pigmentosa?
Retinitis Pigmentosa (RP) is a group of inherited eye diseases impacting the retina positioned at the rear of the eye. This condition is instigated by the demise of photoreceptor cells, the light-signal detecting cells. Presently, no cure for RP exists, and the advancement of fresh remedies hinges on cellular and genetic therapies.
Focus on Rhodopsin-Associated Autosomal Dominant RP (adRP)
The researchers at UCI have concentrated their investigation on a specific molecule with the conviction that it holds the potential to offer a remedy for Rhodopsin-associated autosomal dominant RP (adRP). The molecule in question, Rhodopsin, stands as a pivotal photosensitive molecule in the human retina. It is found within rod photoreceptor cells, and mutations within the Rhodopsin gene constitute a principal cause of adRP.
“More than 150 mutations in rhodopsin can cause Retinitis Pigmentosa, making it challenging to develop targeted gene therapies,” said Krzysztof Palczewski, PhD, Donald Bren Professor, UCI School of Medicine. “However due to the high prevalence of RP, there has been significant investment in research and development efforts to find novel treatments.”
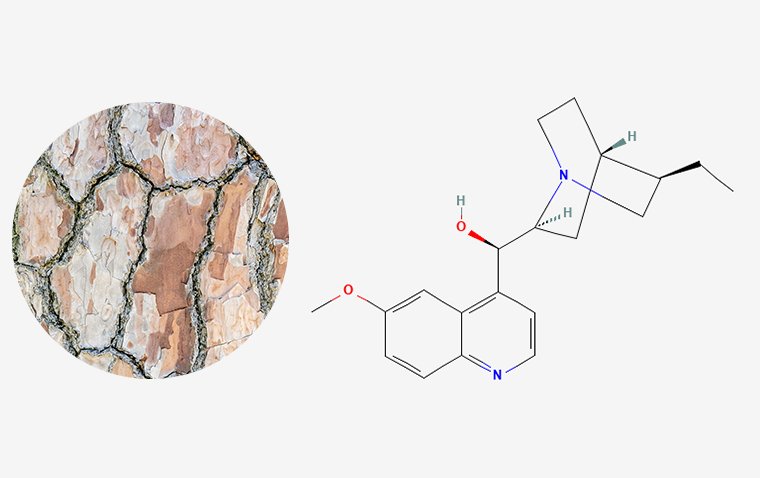
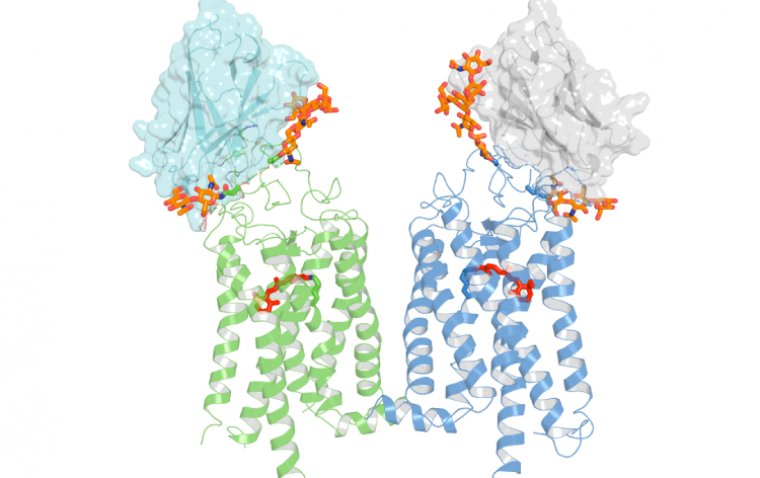
Despite over a century of studying Rhodopsin, crucial intricacies regarding its process of transforming light into a cellular signal have remained challenging to empirically tackle. In this particular investigation, scientists employed a distinctive variety of antibody sourced from llamas, referred to as a nanobody. This nanobody possesses the capability to impede the Rhodopsin photoactivation process, thereby facilitating an in-depth scrutiny at elevated precision.
“Our team has developed nanobodies that work through a novel mechanism of action. These nanobodies have high specificity and can recognize the target rhodopsin extracellularly,” said David Salom, PhD, researcher and project scientist, UCI School of Medicine. “This enables us to lock this GPCR in a non-signaling state.”
The scientists made an intriguing revelation: these nanobodies pinpoint an unforeseen location on the Rhodopsin molecule, situated close to the area where retinaldehyde attaches. Additionally, they observed that the stabilizing influence of these nanobodies could be extended to Rhodopsin mutants linked to retinal diseases, implying their potential application as therapeutic agents.
“In the future, we hope to involve the in vitro evolution of these initial set of nanobodies,” said Arum Wu, PhD, researcher and project scientist, UCI School of Medicine. “We will also evaluate the safety and effectiveness of a future nanobody gene therapy for RP.”
Advancing Nanobody Research for Pre-Clinical Models
The researchers aim to enhance the nanobodies' capacity to distinguish Rhodopsin not only from various species but also from mice, which offer numerous pre-clinical models for adRP. Furthermore, they intend to employ these nanobodies to tackle an enduring objective in the realm of structural elucidation: unraveling the fundamental intermediate stages of Rhodopsin, spanning from its dormant state to the fully ligand-activated state.
Reference
(1).jpg)