FDA Approves Biocon Biologics’ Yesafili™, Interchangeable Biosimilar Eylea
Biocon Biologics today announced that the US FDA has approved the Company’s first-to-file application for Yesafili™ (aflibercept-jbvf), an interchangeable biosimilar aflibercept.
Indications and Uses of YESAFILI
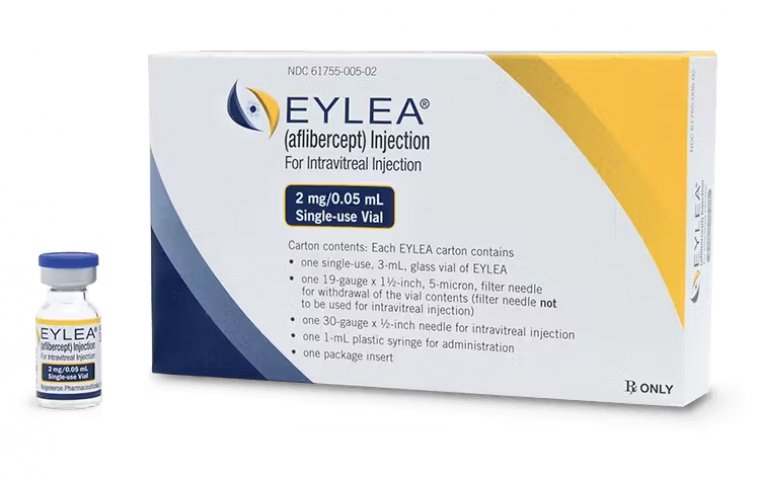
YESAFILI, a vascular endothelial growth factor (VEGF) inhibitor used to treat several ophthalmology conditions, is a biosimilar of its reference product EYLEA® (aflibercept).
YESAFILI is intended for the treatment of neovascular (wet AMD) age-related macular degeneration, visual impairment due to macular edema secondary to retinal vein occlusion (branch RVO or central RVO), visual impairment due to diabetic macular edema (DME), and visual impairment due to myopic choroidal neovascularization (myopic CNV). It is highly similar to the reference product Eylea® (aflibercept). Data shows that YESAFILI has comparable quality, safety, and efficacy to Eylea®.
The approval of YESAFILI marks Biocon Biologics’ expansion into the ophthalmology therapeutic area in the United States following a steady track record of approvals in Europe (September 2023) and the United Kingdom (November 2023), where it was the first biosimilar aflibercept to be approved. The Company has secured a launch date in Canada of no later than July 1, 2025, under the terms of a settlement agreement.
Leadership Remarks on FDA Approval
Shreehas Tambe, CEO & Managing Director, Biocon Biologics Ltd., said: “The FDA approval of YESAFILI (aflibercept) as the first interchangeable biological product to Eylea is a significant milestone for Biocon Biologics marking our entry into Ophthalmology, a new therapeutic area in the United States. YESAFILI is approved for the treatment of neovascular (wet) age-related macular degeneration, macular edema following retinal vein occlusion, diabetic macular edema, and diabetic retinopathy. This approval builds on our successful track record of bringing the first interchangeable insulin, SEMGLEE®, the first biosimilar Trastuzumab, OGIVRI®, and the first biosimilar Pegfilgrastim, FULPHILA®, to patients in the United States.”
Commercial Impact and Market Potential
Matt Erick, Chief Commercial Officer of Advanced Markets, Biocon Biologics Ltd, said: “Biosimilars are crucial for making healthcare more affordable and accessible. YESAFILI will offer ophthalmologists an important new option for patients impacted by macular degeneration and diabetic retinopathy, from a company with a long history of delivering high-quality, science-driven medicines, solely focused on the development and commercialization of biosimilars.”
There are 19.8 million Americans living with age-related macular degeneration (AMD) in the United States. In the U.S., sales of aflibercept were approximately $5.89 billion in 2023.
About Age-related Macular Degeneration (AMD)
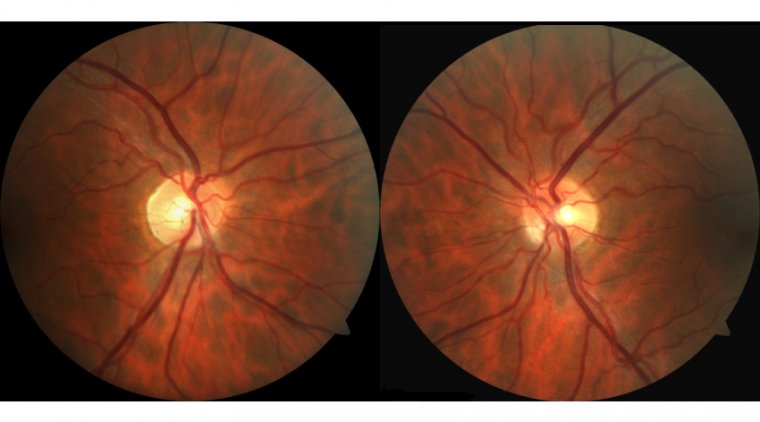
Age-related Macular Degeneration (AMD) is a prevalent eye condition that primarily affects older adults, leading to the deterioration of the macula, the central part of the retina responsible for sharp and detailed vision. As the leading cause of vision loss among people aged 50 and older, AMD significantly impacts daily activities such as reading, driving, and recognizing faces. The condition manifests in two forms: dry AMD, which is characterized by the thinning of the macula, and wet AMD, where abnormal blood vessels grow under the retina, potentially leading to rapid vision loss.
*Stay in the loop and make sure not to miss real-time breaking news about ophthalmology. Join our community by subscribing to OBN newsletter now, and get weekly updates.
(1).jpg)