All About Open-Angle Glaucoma
Open-angle glaucoma is a common type of eye disorder that affects millions of people worldwide. It is also known as primary open-angle glaucoma and is the most common form of glaucoma in the United States. It is a progressive eye disease that can cause permanent vision loss if left untreated. In this article, we will explore what open-angle glaucoma is, what causes it, the symptoms, diagnosis, treatment options, and how it can be prevented.
What Is Open-Angle Glaucoma?
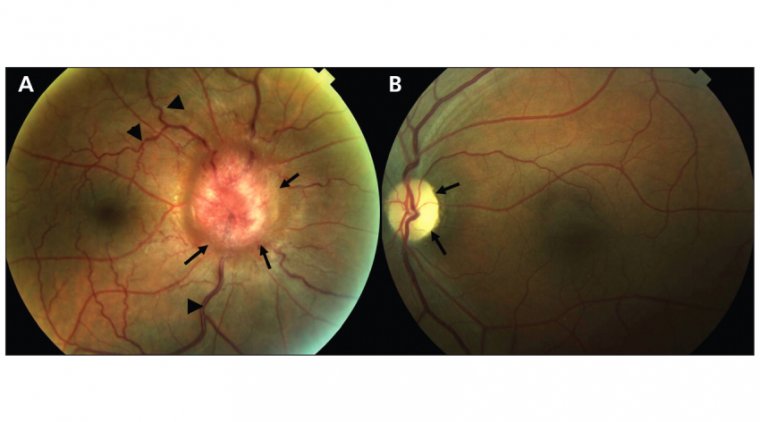
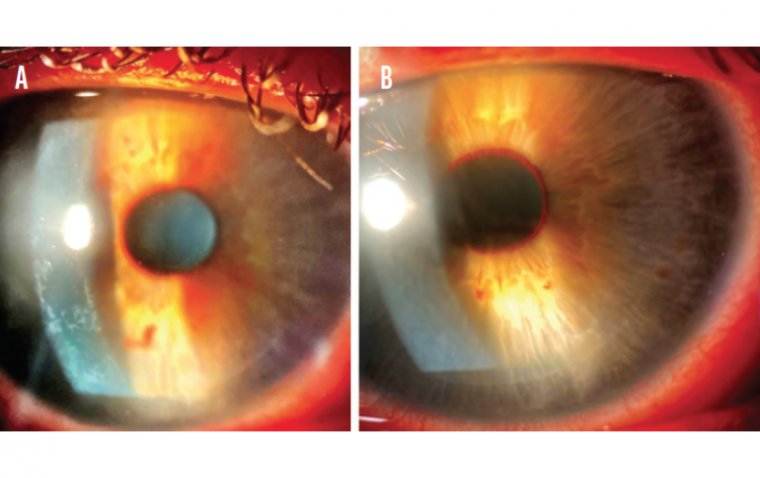
Open-angle glaucoma is a type of eye disease that occurs when the pressure inside the eye increases, leading to damage of the optic nerve. The optic nerve is responsible for carrying visual information from the eye to the brain. In open-angle glaucoma, the drainage system of the eye becomes clogged, leading to an increase in the intraocular pressure. The damage to the optic nerve caused by open-angle glaucoma is irreversible and can result in permanent vision loss.
What Causes Open-angle Glaucoma?
The exact cause of open-angle glaucoma is not fully understood. However, it is believed that a combination of factors can contribute to the development of the disease. These include:
Increased Intraocular Pressure
The pressure inside the eye can increase when the drainage system becomes clogged, leading to damage to the optic nerve.
Age
Open-angle glaucoma is more common in individuals over the age of 40.
Ethnicity
African Americans, Hispanics, and Asians are at a higher risk of developing open-angle glaucoma.
Family History
A family history of open-angle glaucoma can increase the risk of developing the disease.
Medical Conditions
Certain medical conditions such as diabetes and hypertension can increase the risk of developing open-angle glaucoma.
Do Genetics Play A Role In Open-Angle Glaucoma?
Yes, genetics can play a role in open-angle glaucoma. Individuals with a family history of the disease are at a higher risk of developing open-angle glaucoma. Researchers have identified several genes that are associated with open-angle glaucoma, including MYOC, OPTN, and WDR36. However, having a genetic predisposition to the disease does not guarantee that an individual will develop it.
Does Open-Angle Glaucoma Have Any Symptoms?
Open-angle glaucoma is often called the "silent thief of sight" because it can develop slowly and without any symptoms. In the early stages of the disease, there may be no noticeable symptoms. As the disease progresses, individuals may experience:
1. Patchy blind spots in the peripheral or central vision
2. Tunnel vision
3. Difficulty adjusting to darkness
4. Blurred vision
5. Halos around lights
6. Headaches
7. Eye pain
8. Redness in the eye
It is important to note that by the time symptoms appear, significant damage to the optic nerve may have already occurred.
How Is Open-Angle Glaucoma Diagnosed?
Open-angle glaucoma can be diagnosed through a comprehensive eye exam. The exam will include:
Tonometry
This measures the pressure inside the eye (Intra Ocular Pressure.
Visual Field Test
This measures the peripheral vision.
Dilated Eye Exam
This allows the ophthalmologist to examine the optic nerve for signs of damage.
Optical Coherence Tomography (OCT)
This is a non-invasive imaging test that can measure the thickness of the optic nerve and changes in the retinal nerve fiber layer (RNFL).
Treatment Options For Open-Angle Glaucoma
Primary open-angle glaucoma (POAG) is a progressive eye disease that can cause permanent vision loss if left untreated. The management of POAG involves lowering the intraocular pressure (IOP) to prevent further damage to the optic nerve. Treatment options include medication, laser therapy, and surgery.
Medications
Eye drops are the most common treatment for POAG. They work by either reducing the production of fluid in the eye or increasing the drainage of fluid. There are several classes of medications used to treat POAG, including:
Prostaglandin Analogs (e.g. latanoprost, bimatoprost)
These medications increase the outflow of fluid in the eye and are highly effective at reducing IOP.
Beta-Blockers (e.g. timolol)
These medications reduce the production of fluid in the eye and can be used in combination with prostaglandin analogs.
Alpha-Adrenergic Agonists (e.g. brimonidine)
These medications reduce the production of fluid in the eye and can also increase the outflow of fluid.
Carbonic Anhydrase Inhibitors (e.g. dorzolamide)
These medications reduce the production of fluid in the eye and can be used in combination with other eye drops.
Laser Therapy
Laser therapy, also known as trabeculoplasty, is a non-invasive procedure that uses a laser to improve the drainage of fluid in the eye. There are two types of laser therapy used to treat POAG:
Argon Laser Trabeculoplasty (ALT)
This procedure uses a laser to make small burns in the drainage area of the eye to improve fluid outflow.
Selective Laser Trabeculoplasty (SLT)
This procedure uses a laser to stimulate the cells in the drainage area of the eye, which can improve fluid outflow.
Surgery
If medications and laser therapy are not effective in controlling IOP, surgery may be recommended. There are several types of surgery used to treat POAG, including:
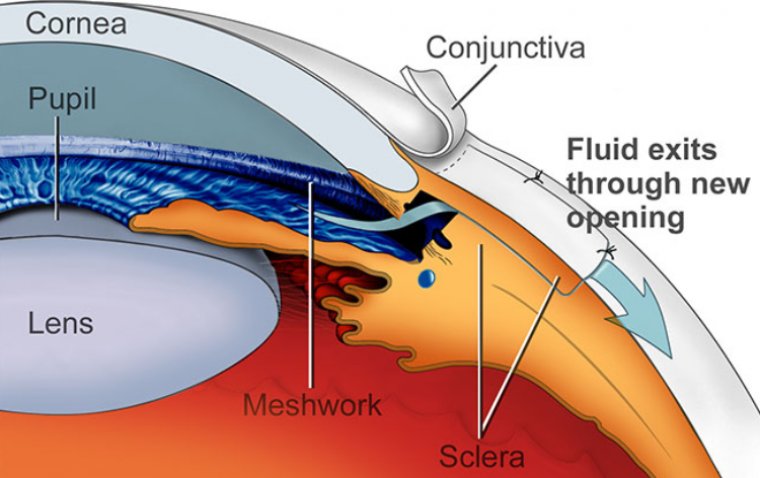
Trabeculectomy
This procedure involves creating a small hole in the eye to allow fluid to drain out of the eye.
Glaucoma Drainage Devices
These are small implants that are placed in the eye to improve fluid drainage.
Minimally Invasive Glaucoma Surgery (MIGS)
These are a group of new surgical procedures that use small incisions and specialized instruments to improve fluid drainage.
It is important to note that the goal of treatment is to lower IOP to prevent further damage to the optic nerve. Treatment does not reverse existing damage. Regular eye exams and monitoring are necessary to assess the effectiveness of treatment and adjust the management plan as needed.
In addition to medical and surgical management, lifestyle changes such as regular exercise, maintaining a healthy weight, and avoiding smoking can also help in the management of POAG. Dietary changes such as reducing caffeine intake and increasing the consumption of green leafy vegetables may also be beneficial.
Comparison Between Medical and Surgical Treatment for POAG
.jpg)
How Can Open-Angle Glaucoma Be Prevented?
Regular Eye Exams
Routine eye exams can detect signs of POAG and allow for early diagnosis and treatment. The American Academy of Ophthalmology recommends a comprehensive eye exam at least once every two years for adults over 40 and more frequently for those with risk factors for glaucoma.
Lifestyle Changes
Maintaining a healthy lifestyle can help reduce the risk of developing POAG. This includes regular exercise, maintaining a healthy weight, and avoiding smoking.
Dietary Changes
Increasing the consumption of green leafy vegetables, which are high in antioxidants and other nutrients, may be beneficial for eye health. Some studies have also suggested that reducing caffeine intake may help to lower intraocular pressure.
Managing Medical Conditions
Certain medical conditions, such as diabetes and hypertension, can increase the risk of developing POAG. Managing these conditions with regular medical care and following recommended lifestyle changes can help to reduce the risk of developing POAG.
Eye Protection
Protecting the eyes from injury is also important in preventing POAG. Wearing protective eyewear during certain activities, such as sports or work that involves exposure to chemicals or flying debris, can help reduce the risk of eye injury.
Conclusion
In conclusion, primary open-angle glaucoma (POAG) is a progressive eye disease that can cause permanent vision loss if left untreated. It is a common condition that affects millions of people worldwide. While the exact cause of POAG is not fully understood, there are several risk factors that can increase the likelihood of developing the disease. Regular eye exams, lifestyle changes, managing medical conditions, and eye protection are some of the ways individuals can reduce their risk of developing POAG or slow its progression if already diagnosed. Treatment options include medication, laser therapy, and surgery. Early diagnosis and treatment are critical in preventing further damage to the optic nerve and preserving vision. It is important to consult with an Ophthalmologist for individualized recommendations on preventing and managing POAG.
FAQ
Open-angle glaucoma is a type of eye disease that can lead to irreversible vision loss. It occurs when the fluid in the eye does not drain properly, leading to an increase in intraocular pressure, which can damage the optic nerve over time.
In the early stages, open-angle glaucoma may not have any symptoms. As the disease progresses, a person may experience vision loss, typically starting with peripheral vision. Other symptoms may include tunnel vision, halos around lights, and eye pain or redness.
Anyone can develop open-angle glaucoma, but some people are at a higher risk than others. Risk factors include being over 60 years old, having a family history of glaucoma, being of African American, Hispanic, or Asian descent, having high intraocular pressure, having thin corneas, and having certain medical conditions such as diabetes and heart disease.