Presbyopia: Understanding Age-Related Vision Impairment
What Is Presbyopia?
Presbyopia is a common, age-related vision condition that affects the eye's ability to focus on near objects. As people age, the lens of the eye becomes less flexible, making it increasingly challenging to adjust focus between objects at different distances. This loss of flexibility leads to a progressive decline in near vision, typically becoming noticeable around the age of 40.
Presbyopia is a natural part of the aging process and affects almost everyone at some point in their lives. It is not a disease or an eye disorder but rather a normal physiological change in the eye's lens. People with presbyopia often find themselves holding reading materials further away or requiring reading glasses or other corrective measures to see up-close objects clearly.
The Science of Presbyopia: Why Age Affects Vision
To understand the science behind presbyopia, it is essential to know how the eye focuses on objects at different distances. The lens, a clear and flexible structure located behind the iris, plays a crucial role in this process. It changes its shape to focus light rays onto the retina, which then transmits the visual information to the brain via the optic nerve.
When focusing on near objects, the lens becomes rounder and thicker, a process called accommodation. This shape change is facilitated by the ciliary muscles, which surround the lens and contract or relax to adjust the lens's curvature.
With age, several changes occur in the eye that result in the development of presbyopia:
1. Lens stiffness: The proteins in the lens undergo age-related changes, causing the lens to lose its elasticity and flexibility. As a result, the lens becomes less capable of changing shape to focus on close objects.
2. Ciliary muscle weakening: The ciliary muscles that control the lens's shape also weaken with age, making it harder for them to contract and facilitate accommodation.
3. Decreased lens support: The lens's supporting structures, such as the lens capsule and the zonules (fibers that hold the lens in place), may also lose their elasticity and strength, further contributing to the diminished focusing ability of the lens.
These age-related changes in the lens and its supporting structures ultimately lead to presbyopia, making it difficult for individuals to see up-close objects clearly without the aid of reading glasses or other corrective measures.
.jpg)
Signs and Symptoms of Presbyopia
Presbyopia typically develops gradually, and its symptoms may initially be subtle. However, as the condition progresses, several signs and symptoms become more apparent. These include:
● Difficulty focusing on near objects: People with presbyopia often struggle to read fine print, thread a needle, or perform other close-up tasks without straining their eyes.
● Holding reading materials at a distance: One of the most common signs of presbyopia is the need to hold books, newspapers, or digital devices further away to see the text clearly.
● Eye strain or fatigue: When trying to focus on near objects, individuals with presbyopia may experience eye strain, discomfort, or fatigue, particularly after prolonged close-up work.
● Headaches: Difficulty focusing on close objects may cause headaches, especially after extended periods of reading or engaging in other near-vision tasks.
● Difficulty adjusting focus: People with presbyopia might find it challenging to switch focus between near and distant objects, such as reading a menu and then looking at a distant street sign.
● Need for increased lighting: As presbyopia progresses, individuals may find that they require brighter light to see up-close objects more clearly, such as when reading or working on intricate tasks.
Managing Presbyopia
Managing presbyopia typically involves the use of corrective measures to improve near vision. While presbyopia cannot be reversed or cured, several options are available to help individuals see up-close objects more clearly. These include:
1. Reading glasses: Over-the-counter or prescription reading glasses can help individuals with presbyopia see near objects more clearly. The strength of the glasses, measured in diopters, can be adjusted to match the individual's specific needs.
2. Bifocals, trifocals, or progressive lenses: For those who already wear prescription glasses for distance vision, bifocals, trifocals, or progressive lenses can be used to correct both distance and near vision. These lenses have different zones for various viewing distances, allowing individuals to see clearly at all ranges without switching glasses.
3. Contact lenses: Multifocal or monovision contact lenses are also available for individuals with presbyopia. Multifocal lenses have different zones for near and distance vision, while monovision lenses involve wearing a contact lens for distance vision in one eye and a contact lens for near vision in the other eye.
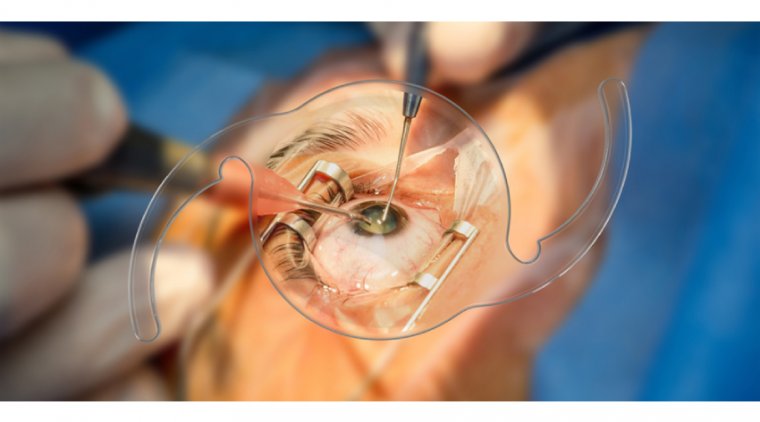
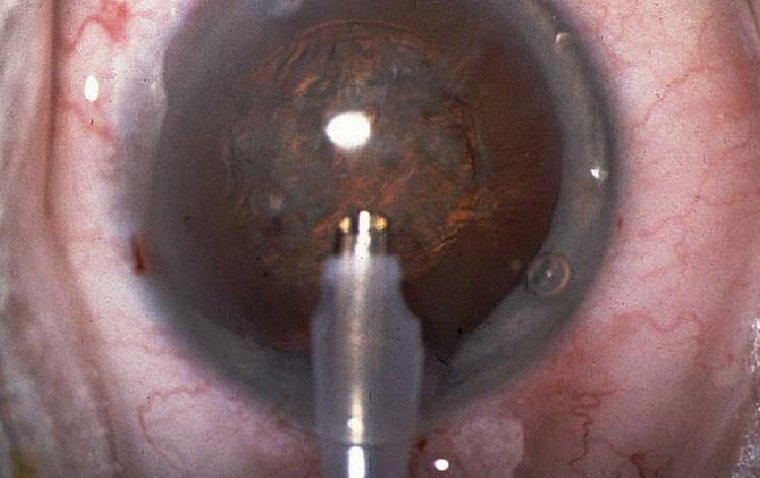
4. Refractive surgery: Surgical procedures like LASIK or PRK can be modified to address presbyopia using a technique called monovision, similar to monovision contact lenses. However, not all individuals with presbyopia are suitable candidates for refractive surgery, and the procedure may not entirely eliminate the need for reading glasses.
5. Corneal inlays: This minimally invasive procedure involves implanting a small, ring-shaped device into the cornea of the non-dominant eye to improve near vision. Corneal inlays are removable and can be replaced if necessary.
Corrective Lenses: Glasses and Contacts
Corrective lenses, including glasses and contact lenses, are the most common and non-invasive solutions for managing presbyopia. They work by compensating for the eye's decreased ability to focus on near objects, allowing individuals to see clearly at various distances. Here's a brief overview of the different types of corrective lenses available for presbyopia:
● Reading glasses: These are simple, over-the-counter glasses that help individuals focus on close-up tasks, such as reading or sewing. Reading glasses come in various strengths, measured in diopters, and can be easily purchased at pharmacies, supermarkets, or online retailers.
● Prescription glasses: For those who require correction for both distance and near vision, prescription glasses can be customized with bifocal, trifocal, or progressive lenses. Bifocal lenses have two distinct zones: the upper part for distance vision and the lower part for near vision. Trifocal lenses have three zones: distance, intermediate, and near vision. Progressive lenses provide a seamless transition between distance, intermediate, and near vision zones without visible dividing lines.
● Multifocal contact lenses: These contact lenses are designed with multiple zones for different viewing distances, providing a smooth transition between near and distance vision. Multifocal contact lenses can be either soft or rigid gas permeable, and they are available in various designs, such as concentric, aspheric, or segmented.
● Monovision contact lenses: This approach involves fitting one eye with a contact lens for distance vision and the other eye with a contact lens for near vision. The brain learns to prioritize the input from each eye depending on the viewing distance, allowing for clear vision at both near and far ranges.

Surgical Options: LASIK and Other Procedures (New eye drops)
Surgical options for treating presbyopia aim to correct the eye's focusing ability or alter the cornea's shape to improve near vision. These procedures may not be suitable for everyone, and the need for reading glasses may not be entirely eliminated. Some of the surgical options available for presbyopia include:
1. Monovision LASIK: This laser eye surgery modifies the traditional LASIK procedure to create monovision, where one eye is corrected for distance vision and the other for near vision. The brain learns to prioritize the input from each eye depending on the viewing distance, providing clear vision at both ranges.
2. Photorefractive keratectomy (PRK): Similar to LASIK, PRK can also be adjusted to create monovision. PRK involves removing the outer layer of the cornea and reshaping the underlying tissue with a laser, allowing the cornea to refocus light onto the retina properly.
3. Conductive keratoplasty (CK): CK uses radiofrequency energy to reshape the cornea and improve near vision. The procedure involves applying heat to specific points on the peripheral cornea, causing it to shrink and steepen, which enhances near vision.
5. Corneal inlays: Corneal inlays involve implanting a small, ring-shaped device into the cornea of the non-dominant eye to improve near vision. This minimally invasive procedure can be reversed, and the inlay can be replaced if necessary.
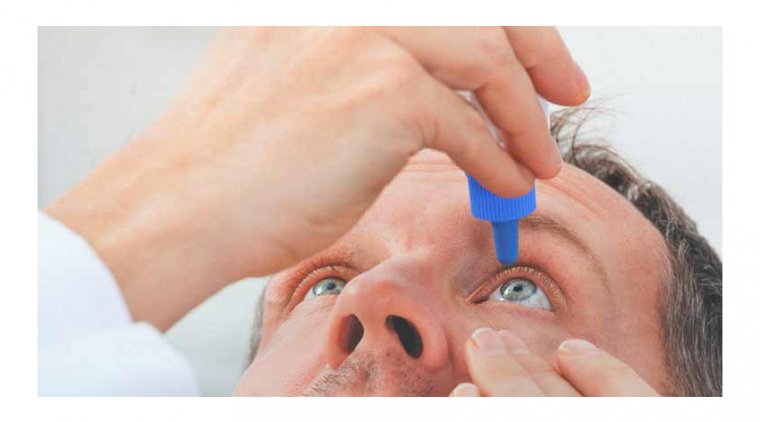
6. New eye drops: Emerging treatments like eye drops that temporarily improve near vision are being developed. For instance, the FDA has approved Vuity (pilocarpine hydrochloride ophthalmic solution) for the treatment of presbyopia. The eye drops work by constricting the pupil, which increases the depth of focus and improves near vision. However, the effects of the drops last only a few hours, and they may cause side effects such as headache, eye irritation, or blurred distance vision.
Lifestyle Changes to Improve Vision
While presbyopia is a natural part of the aging process and cannot be prevented, certain lifestyle changes can help maintain overall eye health and potentially slow down the progression of vision decline. Here are some recommendations for promoting better vision:
● Balanced diet: Consuming a diet rich in fruits, vegetables, whole grains, and lean proteins can support eye health. Nutrients like vitamin A, vitamin C, vitamin E, zinc, lutein, and zeaxanthin are essential for maintaining good vision and reducing the risk of eye diseases.
● Regular exercise: Engaging in physical activity can help improve blood circulation, lower blood pressure, and maintain a healthy weight, all of which contribute to better eye health. Aim for at least 150 minutes of moderate-intensity aerobic activity or 75 minutes of vigorous-intensity aerobic activity per week.
● Proper lighting: Ensure your reading and work areas have adequate lighting to reduce eye strain. Consider using task lighting or adjustable lamps that allow you to control the brightness and direction of the light.
● Breaks and eye exercises: When reading or working on a computer, follow the 20-20-20 rule: every 20 minutes, take a 20-second break and look at something 20 feet away. This helps reduce eye strain and fatigue. Additionally, practice eye exercises such as focusing on near and far objects and performing eye movements in different directions.
● UV protection: Wear sunglasses that block 100% of UVA and UVB rays to protect your eyes from the harmful effects of the sun. A wide-brimmed hat can also provide extra protection during outdoor activities.
● Regular eye exams: Schedule regular eye exams to monitor your vision and overall eye health. Early detection of vision changes or eye conditions can lead to more effective treatments and better outcomes.
Avoid smoking: Smoking increases the risk of various eye diseases, including cataracts, macular degeneration, and optic nerve damage. Quitting smoking can reduce these risks and promote overall health.
.jpg)
Looking Ahead: Advances in Presbyopia Management and Treatment
As the global population continues to age, the demand for innovative solutions to manage and treat presbyopia is on the rise. Researchers and eye care professionals are continuously exploring new technologies and therapies to address this common age-related vision problem. Some of the promising advancements in the field of presbyopia management and treatment include:
1. Advanced multifocal intraocular lenses (IOLs): Newer generation multifocal IOLs offer improved visual outcomes and fewer visual disturbances, such as glare and halos, compared to earlier models. These lenses are implanted during cataract surgery or refractive lens exchange, providing patients with a more comprehensive range of vision.
2. Scleral implants: Scleral implants are small devices inserted into the white part of the eye (sclera) to stretch and reshape the cornea, improving near vision. These implants are in the experimental stage and may offer a less invasive alternative to corneal inlays.
3. Presbyopia-correcting eye drops: As mentioned earlier, the FDA has approved Vuity (pilocarpine hydrochloride ophthalmic solution) for the treatment of presbyopia. Researchers are also working on other eye drops that target the lens of the eye, aiming to restore its elasticity and focusing ability.
4. Adjustable IOLs: Researchers are developing intraocular lenses that can be adjusted after implantation using light or other noninvasive methods, allowing for customized vision correction without the need for additional surgery.
5. Stem cell therapy: Scientists are exploring the potential of using stem cells to regenerate the eye's natural lens, which could provide a more permanent solution for presbyopia. While still in the early stages of research, this approach holds promise for the future of vision correction.
To Sum Up...
Presbyopia is a natural, age-related vision change that affects millions of people worldwide. As we age, our eyes' lenses lose their flexibility, making it increasingly difficult to focus on close objects. Understanding the signs and symptoms of presbyopia is crucial for seeking appropriate corrective measures and maintaining good vision and overall eye health.
A variety of management options are available for presbyopia, including corrective lenses, surgical interventions, and lifestyle changes that promote healthy eyes. Staying up-to-date with the latest advancements in presbyopia treatment can help individuals make informed decisions about their eye care.
In conclusion, it is essential to recognize the signs of presbyopia, schedule regular eye exams, and consult with an eye care professional to determine the best course of action for your unique vision needs. By adopting a proactive approach to managing age-related vision changes, you can maintain a high quality of life and continue to enjoy the world around you, one focused moment at a time.
(1).jpg)