The Basics of Excimer Laser
In the field of ophthalmology, laser technology is used in many basic and clinical disciplines and specialities. It has played an important role in promoting the development of ophthalmology.
Advancements in technology have allowed improvements in the surgical safety, efficacy, speed and versatility of the laser, especially in corneal refractive surgery.
Because of the increasing numbers of applications in ophthalmology and their successful implementations, ophthalmic use of laser technology is expected to continue flourishing.
Excimer Laser Principle of Work
The excimer laser is based on the combination of two gases: a noble gas and halogen. Both of these are generally stable in their normal low-energy state.
When a high-voltage electrical discharge is delivered into the laser cavity containing these gases, the gases combine to form a higher energy excited-gas state compound. The term “excimer” is derived from a contraction of “excited dimer”.
On the dissociation of this high-energy compound, a photon of energy is released that corresponds to the bond energy of the noble gas-halogen molecule. This wavelength of light energy is amplified in the laser system, resulting in the production of a discrete high energy pulse of laser energy.
The specific wavelength of an excimer laser depends on the composition of the gases used in the laser system. Excimer laser systems in current clinical use rely on argon and fluorine gases. The argon-fluorine excimer lasers emit energy at a wavelength of 193 nm.
This wavelength falls in the UV-C range of the light spectrum. In contrast, the krypton-fluoride excimer laser used in early laboratory studies emits a wavelength of 248 nm.
Laser energy at 193 nm is very well absorbed by the proteins, glycosaminoglycans and nucleic acids comprising the cornea because of its sufficient photon energy (6.4 eV) and precision (only penetrating the superficial layer; 0.3 μm).
The tissue-ablation depth is positively correlated with the logarithm of laser density; 1-J/cm2 energy can ablate approximately 1-μm of corneal tissue.
Since 193 nm photon is of higher energy than the molecular bond strength of these compounds, absorption of the laser energy results in breaking of the bounds. The resulting molecular fragments are ejected from the surface of the cornea at supersonic speeds.
The goal is to reshape the cornea so that rays of light that enter the eye are focused clearly onto the retina. It is important to understand that the excimer laser does not cut tissue like a scalpel; rather it ablates or removes tissue from the corneal surface.
The ablated material appears as an effluent plume that upon analysis has been shown to consist of a variety of high-molecular-weight hydrocarbons.
There is a concern about the potential for mutagenesis or carcinogenesis with any laser radiation, especially in the ultraviolet light spectrum.
You can’t work long in eye care without becoming aware of excimer laser procedures. Excimer lasers are the tool that enabled the explosion in laser vision correction procedures in the 1990s.
According to the Refractive Surgery Council, laser vision correction surgeries increased 48% year-over-year from Q3 2020 to Q3 2021 — the vast majority of those procedures performed with an excimer laser.
So, given its primacy in vision correction, ophthalmic professionals need to understand the technology and how to present it to patients.
A little history - Excimer laser technology was developed by IBM in 1976 and was used extensively in the creation of computer microchips.
In ophthalmology, the excimer was first used clinically in 1985. In 1988, Dr. Marguerite McDonald verified the theoretical basis of photorefractive keratectomy (PRK) in a series of animal experiments that led to the first successful application in a normal sighted eye in May 1988.
As Brian S. Boxer Wachler, MD, explains on All About Vision (bit.ly/3rRGRVg), the excimer laser is a cold laser, which differentiates itself from other lasers we use in ophthalmology (such as the Argon laser, the Yttrium-Aluminum Garnet — best known as the YAG laser — and the femtosecond).
It emits a cool beam of ultraviolet light of a specific wavelength to precisely remove corneal tissue. When the surface of the cornea is reshaped in the appropriate manner, it allows light rays to focus properly onto the retina for clear vision.
The FDA approved the excimer laser for use in PRK in 1995; today, it is used to correct myopia, hyperopia and astigmatism by reshaping the cornea after removing the very top layer of corneal cells (the epithelium) with a surgical instrument or an alcohol solution.
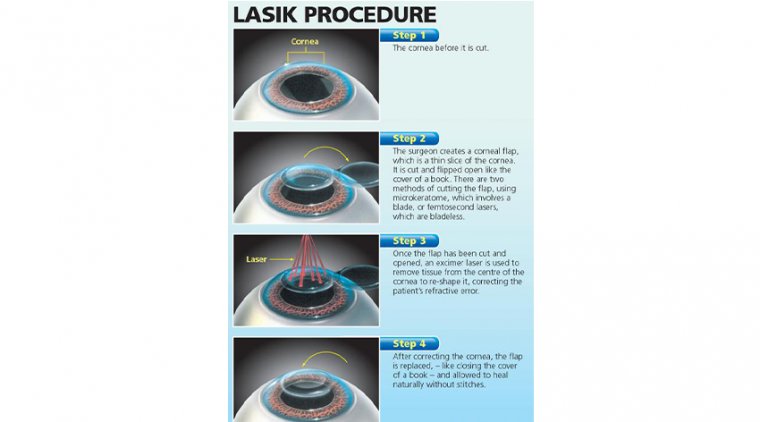
In 1998, the FDA approved laser-assisted in-situ keratomileusis (LASIK), a procedure that calls for creating a flap in the corneal tissue of approximately 120 to 130 microns, reshaping the cornea with the laser, and then repositioning the flap on the cornea.
LASIK quickly became the more requested vision correction procedure as it has a much faster recovery time than PRK and delivers immediate results to patients.
Now we will review the key pieces a practice needs in place to successfully incorporate excimer laser procedures.
Appoint a refractive coordinator - First, we recommend that your practice assign one technician as the refractive coordinator. This person works directly with the refractive surgeon in the clinical setting, providing support.
This support includes screening patients, performing preliminary workups, scheduling procedures at the laser center, and scheduling postoperative checkups. The refractive coordinator is the go-to person for refractive patients and for office staff.
Patient selection - Patients seeking LASIK today undergo a thorough ophthalmic examination to determine if they are good candidates.
Prior to coming to our facility for their first appointment with the ophthalmologist, we schedule them at our laser center for preliminary measurements and screening.
We perform wavefront and anterior segment tomography (Oculus, Pentacam) measurements to determine refraction and corneal thickness.
The range of correction for LASIK and PRK is approximately +5.00 D to −10.00 D sphere; astigmatism between −0.25 D and −6.00 D may also be corrected. Anterior segment tomography, among other things, measures corneal thickness, which is critical to the discussion of whether to perform LASIK or PRK.
The cornea must be more than 500 microns thick to allow for the creation of the flap and the removal of tissue to correct the refractive error. Leaving the cornea too thin would put the patient at risk for serious complications.
Corneas that measure less than 500 microns are better suited to PRK, as no flap is created in that procedure. Studies report the visual results of PRK vs. LASIK at 6 months postop are very similar. The major difference is the initial healing during the first few days.
Contraindications - Some patients are simply not good candidates for laser vision correction. Factors that would put a patient in this category are unstable refractive error, corneal warpage, keratoconus, corneal disease, cataract, retinal/macular disease, and systemic conditions such autoimmune diseases.
Additionally, a thin cornea can cause a LASIK-seeking patient to instead qualify for PRK. With careful counseling before surgery, a patient can go forward with PRK and have as good an outcome as one who had LASIK.
Make sure the patient understands that PRK is uncomfortable in the immediate postoperative period and vision takes days to clear.
Pre-surgery prep - We instruct patients to discontinue soft contact lens wear for a minimum of 3 days prior to evaluation.
Prior to the laser treatment, we recommend they discontinue contact lens wear for at least 2 weeks. However, the time needed to discontinue contact lens wear can be longer than that — it depends solely on the how stable the cornea appears.
For example, a patient who has been wearing rigid gas permeable (RGP) contact lens will require much longer discontinuation of wear, because this type of lens tends to mold or reshape the cornea. Unmolding is imperative to a good outcome of laser surgery because the patient’s cornea changes shape as it unmolds.
If the cornea is still changing shape and PRK or LASIK is performed before stabilization, then the outcome cannot be predicted.
The refractive coordinator may have the RGP wearer come in for several refractions prior to the preliminary examination and give the patient a temporary prescription for glasses.
The refractive coordinator will explain to the patient that they may have to come back to the office every time their vision deteriorates enough to require a change in glasses. This can take up to 1 month for every decade of RGP wear before the practice can obtain an accurate refraction.
At each visit to the office, the patient will need a corneal topography (CT) measurement. Once two consecutive CT measurements show no further changes and the refraction is stable, the patient is ready for a complete ophthalmic examination that will include a cycloplegic refraction (See "Refractive Evaluation Steps").
Your final step - A last, critical requirement in educating patients about refractive laser surgery: The patient must have realistic expectations of the procedure.
This can be the most important and difficult part of the evaluation. Here, being honest and not over promising visual acuity results can help lead to greater patient satisfaction with the procedure.
(1).jpg)