Diagnosis and Treatment of Corneal Ulcers
Corneal ulcers may be caused by infections with bacteria, fungi, viruses, or parasites such as Acanthamoeba (which lives in contaminated water).
Ulcers may begin with a corneal injury, such as with severe dry eyes, if a foreign object scratches, penetrates, or lodges in the eye or, more often, if the eye is irritated by a contact lens, especially when contact lenses are worn during sleep or are not adequately disinfected.
Viral corneal ulcers (often due to herpesvirus) can be triggered to recur by physical stress or may recur spontaneously. A deficiency of vitamin A and protein may lead to the formation of a corneal ulcer.
When the eyelids do not close properly, the cornea may become dry and irritated. This kind of irritation can lead to injury and the development of a corneal ulcer. Corneal ulcers may also result from in-growing eyelashes (trichiasis), an in-turned eyelid (entropion), or eyelid inflammation (blepharitis).
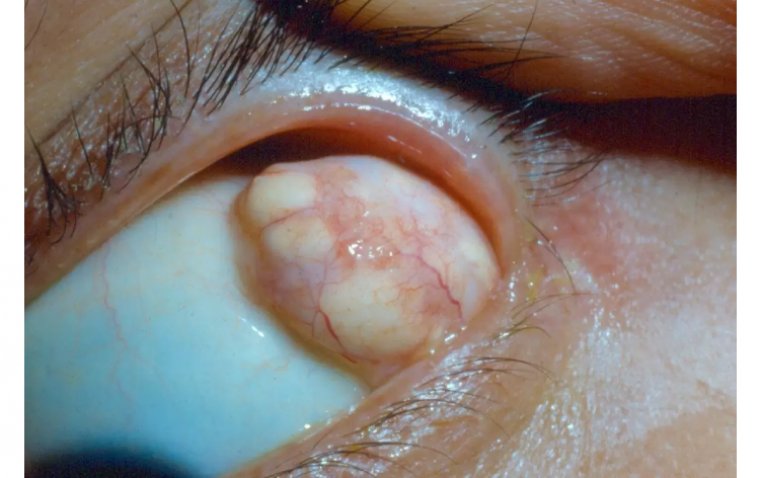
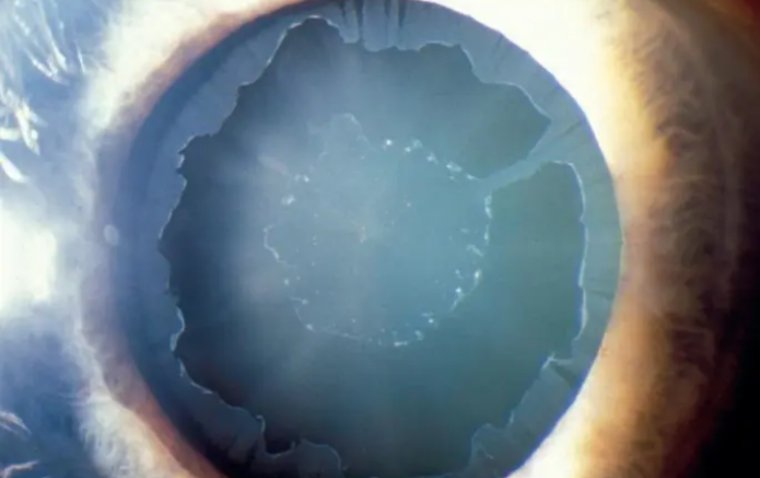
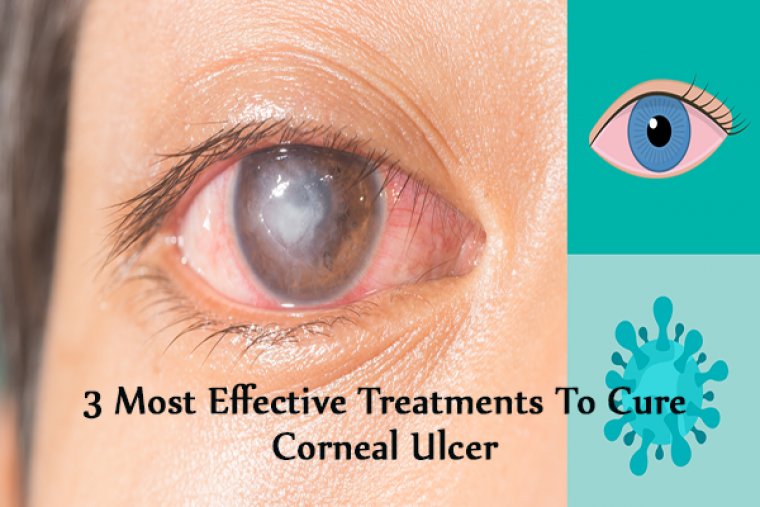
Corneal ulcer is an inflammatory or, more seriously, infective condition of the cornea involving disruption of its epithelial layer with involvement of the corneal stroma.
It is a common condition in humans particularly in the tropics and the agrarian societies.
In developing countries, children afflicted by Vitamin A deficiency are at high risk for corneal ulcer and may become blind in both eyes, which may persist lifelong.
A corneal ulcer usually refers to having an infectious cause while the term corneal abrasion refers more to physical abrasions.
We have all experienced that feeling of “impending doom” when examining the emergency walkin corneal ulcer. This is because many corneal ulcers progress rapidly and can be vision or eye threatening.
As the pandemic has demonstrated, countless microorganisms exist. Fortunately, when dealing with the cornea and ocular surface, there is no need to be concerned with a vast majority of these organisms.
The most common corneal infections in the United States are bacterial (related to contact lens wear) and viral (such as herpes simplex virus [HSV] or herpes zoster virus [HZV]). Here, we explain how to correctly diagnose corneal ulcers and treat the various causes of ulcers.
Diagnosis Overview
Because these corneal infections can be eye and/ or vision threatening, making the correct diagnosis is paramount.
To effectively diagnose and treat these infections, it is important to separate the possible infectious organism into four categories: bacterial, fungal, parasitic and viral.
Although a presumptive diagnosis may be helpful for early treatment, a thorough history, examination and work-up are imperative to identify the etiology of the ulcer and achieve a successful resolution.
Upon presentation, making the correct diagnosis must start with patient history. For example:
• Do they wear contact lenses? (Think about gram-negative bacteria like pseudomonas).
• Did they get poked in the eye with a tree branch? (Look for fungal keratitis).
• Do they swim or shower in their contacts? (Acanthamoeba should be high on your differential list).
• Are they atopic? (Consider HSV) Beyond the slit lamp exam, talk with the patient and examine them outside the slit lamp.
Also, utilize the history and exam to formulate a thorough differential diagnosis, considering all possible causes — regardless of rarity — and determine which diagnoses are more likely based upon the information gathered.
Still, initial treatment may need to be broad spectrum until the diagnosis is confirmed. One can easily be fooled, and an incorrect diagnosis may be devastating for the eye and vision.
Bacterial Keratitis
Bacterial keratitis is often vision and eye threatening because of its rapidly progressive and destructive course. Risk factors include:
• Contact lens wear (with a change in ocular flora and micro abrasions to the epithelium)
• Trauma (abrasions, erosions, lacerations, chemical and thermal injuries)
• Surgery (healing epithelial defects and exposed sutures)
• Comorbid conditions (HSV, HZV or neurotrophic keratitis with defects)
• Chronic immunosuppression (corneal transplants with chronic topical steroid use)
Bacterial ulcers are separated into gram-positive and gram-negative infections. Although they usually present with some differences on examination, a work-up is often required to determine the exact culprit.
For example, gramnegative infections, such as pseudomonas species (commonly seen in contact lens-related ulcers) commonly have more collagenase activity with adherent mucous, corneal thinning and possible melting.
Despite assumptions as to the type of bacteria, broad coverage therapy is recommended until you identify the bacteria. Ideally, most patients should be cultured and started on broad-spectrum, fortified antibiotics.
However, this may not always be feasible, financially or practically. According to the AAO practice patterns, most ulcers can be treated with empiric therapy without smears or cultures.
However, cultures and fortified antibiotics are recommended for ulcers that are large (usually greater than 2 mm), central, associated with melting or thinning, chronic or unresponsive to broad spectrum antibiotics, and associated with corneal surgery.
Fortified antibiotics should include a broad spectrum regimen of drops, such as cefazolin 25-50mg/ml and tobramycin or gentamicin 9-14mg/ml every hour while awake.
An alternative regimen, including vancomycin 25-50mg/ml and ceftazidime 25-50mg/ml, could be considered, especially for partially treated or unresponsive ulcers.
The combinations of antibiotics may be varied based on sensitivities and allergies (ie, penicillin allergy). Subconjunctival injections can be considered.
Treatment for small, peripheral, non-vision threatening ulcers with no thinning may include broad-spectrum commercially available antibiotic drops such as the later generation fluoroquinolones (moxifloxacin, besifloxacin, gatifloxacin) and bacitracin or erythromycin ointment.
Based on our experience, we recommend the following pearls to manage bacterial ulcers:
• Act aggressively. If in doubt, you will never be criticized for culturing, using fortified antibiotics and following the patient too closely.
• If you suspect gram-negative bacteria, use double coverage. Always treat the ulcer with broad-spectrum antibiotics until you identify the organism. However, if you suspect a gram-negative, such as pseudomonas in a contact lens-related ulcer (collagenase, adherent mucous, melting, etc.), add a second treatment to the fortified tobramycin or ceftazidime, such as ciprofloxacin.
• Use caution with topical steroids. It may be prudent to wait on topical steroids until the ulcer is epithelialized or you are convinced that the ulcer has been sterilized.
Adjunctive topical steroids may be associated with improved clinical long-term outcomes for bacterial ulcers not caused by Nocardia. Steroids may have the benefit of lessening vision loss due to scarring.
However, certain bacteria, such as pseudomonas and Streptococcus pneumoniae, may thrive again with initiating topical steroids too early. In addition, steroids may increase collagenase activity and melting.
• When in doubt, send it out. You will never be criticized for sending an ulcer to a cornea specialist. If the patient is to be seen that day, fight the urge to use an antibiotic drop before sending the patient to the specialist.
This may lower the yield if cultures are to be done. If the patient cannot be seen until the next day or later, then start broad-spectrum antibiotics as indicated.
Fungal Keratitis
Fungal infections of the cornea are more common in developing countries. Fungal keratitis represents less than 5-10% of corneal infections in the United States, and they are more common in the south due to warmer, humid weather.
Common risk factors include:
• Trauma (especially with vegetative matter such as a tree branch, plant or soil)
• Contact lens wear
• Immunosuppression
• Corneal surgery (especially corneal transplantation). Fungal cornea infections may also be a secondary infection in conjunction with other infections such as HSV and VZV.
With fungal keratitis, the organisms can penetrate an intact Descemet’s membrane and enter into the anterior chamber and vitreous. This means there is a higher likelihood that a fungal keratitis with a hypopyon represents an endophthalmitis rather than a corneal ulcer with a sterile hypopyon.
Fungal keratitis must be considered when patient history indicates at least one of the risk factors listed above. Although they may have an appearance similar to bacterial corneal ulcers, fungal infections may have less inflammation with less corneal edema and less conjunctival injection.
Other differences include no epithelial defect, a deeper infiltrate (especially with a clear stroma overlying it), a ring infiltrate (this may also be seen with acanthamoeba infections), an infiltrate with irregular, feathery edges, an endothelial plaque or satellite lesions.
As mentioned, presence of a hypopyon could represent intraocular involvement. Yeast infections, such as Candida, may also present with superficial, white, raised lesions. After an examination and assessment of the risk factors, confirmation of a fungal keratitis diagnosis is usually determined by corneal scraping and the use of specific stains and cultures (ie, Sabouraud’s media).
Fungal elements in corneal infection can be identified via a biopsy, polymerase chain reaction (PCR) and confocal microscopy to assess the presence of branching filaments. Fungal keratitis treatments include topical antifungals.
In addition, systemic treatments may be needed for deeper infiltrates, severe infections, scleral involvement or possible endophthalmitis. Topical amphotericin B (0.15%- 0.30%) is the preferred agent against yeasts, such as Candida, as well as some filamentous fungus caused by Aspergillus.
This medication must be compounded. Topical natamycin 5% (Natacyn, Eyevance Pharmaceuticals) is efficacious against most filamentous fungi such as Fusarium. Patients use these topical treatments every 1-2 hours and often together as broad spectrum for initial treatment, especially prior to confirmation of the type of fungus and for cases of Aspergillus.
Voriconazole 1% is another topical treatment for patients who do not respond to other therapies. This medication also needs to be compounded. The Mycotic Ulcer Treatment Trial (MUTT I) demonstrated that treatment with natamycin was associated with better outcomes than topical voriconazole for filamentous fungal keratitis.
This evidence has led to use natamycin topically for filamentous fungal infections at the beginning of treatment followed by an addition of topical voriconazole as necessary. Epithelial keratectomy and debridement may help with penetration and efficacy of the topical therapies.
Other medications include systemic antifungals, such as ketoconazole (200-600 mg/day), fluconazole (200-400 mg/day), itraconazole (200mg/ day) and voriconazole (200-400 mg/day). Oral voriconazole is another consideration due to its excellent intraocular penetration and broad coverage.
In MUTT II, oral voriconazole showed no difference vs placebo in treating fungal keratitis, except for a sub-group of Fusarium ulcers, where there was a benefit in outcome for the oral voriconazole group. For severe or poorly responsive fungal ulcers, additional treatment options include corneal cross-linking (CXL) or therapeutic penetrating keratoplasty.
Management of fungal keratitis often requires multiple therapies for many months to avoid recurrence.
Parasitic Infections
Parasitic infections of the cornea are as difficult to diagnose and as complex to treat as fungal keratitis. About 90% of reported cases of Acanthamoeba keratitis are associated with contact lens use and are bilateral in an about 7% to 11% of cases.
However, non-contact lens wearers can get Acanthamoeba from trauma or exposure to water or soil. Risk factors include swimming or showering in contact lenses, rinsing or storing contact lenses in tap water or homemade saline and rinsing or lubricating contact lenses in the patient’s mouth.
Patients with Acanthamoeba often experience pain out of proportion to findings as well as photophobia, foreign body sensation and decreased vision. These patients can present with a protracted and progressive course with no improvement on antibiotics or antivirals due to it being misdiagnosed as HSV and VZV, causing a delay in the true diagnosis by weeks or months.
Treatment with topical steroids may delay or worsen the course as well. Because it is uncommon, an Acanthamoeba diagnosis must be considered in the first place. On clinical examination, corneal findings may include punctate epitheliopathy (often diffuse), a dendrite-like staining and a corneal infiltrate, which is nonsuppurative but may have a secondary bacterial infiltrate associated with it.
A ring infiltrate occurs 50% of the time and should trigger a consideration of an Acanthamoeba diagnosis. The differential diagnosis for a ring infiltrate, in addition to Acanthamoeba, includes fungal keratitis, an immune ring in some viral keratitis and topical anesthetic abuse.
An endothelial plaque and corneal thinning may be associated with the infiltrate, and the patient may have a haze or infiltrate surrounding the corneal nerves (radial perineuritis). Prolonged, protracted cases may develop scleral involvement, leading to a focal, nodular or diffuse scleritis.
This forebodes a poor prognosis. Once Acanthamoeba is suspected, you can confirm a diagnosis in several ways. First, a corneal scraping can be done for staining and culture.
The cultures should be performed on a nonnutrient agar with an E coli or Enterobacter aerogenes overlay or a buffered charcoal-yeast extract agar. These plates may not be available to you, so the scraping can be placed in Page’s saline for transport to a lab to be plated with the above-mentioned agars.
In addition, culture the contact lens and/or the contact lens case for Acanthamoeba if suspicion is high and you have a negative culture as false negatives can occur. Other diagnostic tools include PCR detection, confocal microscopy and corneal biopsy. Early diagnosis aids the prognosis.
Treatment is difficult secondary to the cyst formation, which can withstand many treatments, and treatment of a year or more is often required. Multi-treatment is often required and recommended due to the cyst formation.
Options include topical and oral antiamebic therapy, lamellar and penetrating keratoplasty and CXL. Steroids are usually avoided but may be beneficial once anti-amebics are on board. Acanthamoeba medications can be separated into four categories:
• Biguanides. These are used topically and consist of polyhexamethylene biguanide and chlorhexidine. Biguanides must be in the treatment regimen as they may be the best option to be cysticidal. If monotherapy is considered, the biguanides should be the drugs of choice.
• Diamidines. These consist of propamidine isethionate (0.1%) (Brolene, Sanofi), dibromopropamidine (0.15%) and hexamidine (0.1%). Brolene and hexamidine are the diamidines most commonly used but are difficult to obtain (Brolene is available in Europe).
• Aminoglycosides. These include neomycin and paromomycin topically.
• Imidazoles/triazoles. These may be topical and/or oral and include voriconazole, miconazole, clotrimazole, ketoconazole and itraconazole. The common protocol for Acanthamoeba treatment includes biguanide in addition to one, two or all three of the remaining options.
The number of meds and frequency are dependent upon severity, toxicity and response. Other amoebic infections of the cornea, fortunately, are rare. Microsporidiosis is a lesser-known parasitic infection of the cornea.
Microsporidia are intracellular protozoa that can infect the cornea of both immunocompetent (stromal keratitis) and immunosuppressed patients (conjunctivitis and superficial epithelial keratopathy).
The stromal infiltrate are seen in immunocompetent individuals, while the keratoconjunctivitis variant is usually seen in AIDS patients. Diagnosis is made by scraping for light microscopy using stains such as Geimsa, 1% acid fast or Brown and Hopps stain or by transmission electron microscopy.
Cultures may be difficult as these organisms are fastidious. There is no definitive treatment, but topical fumagillin is suggested, especially for the keratocon junctivitis. Reported cases have suggested using propamidine.
Viral Keratitis
Viral keratitis may be the most common of the corneal infections. It is usually caused by HSV or, to a lesser extent, VZV. Viral keratitis may masquerade as several other corneal diseases but should always be considered, especially for patients with a history of atopic disease.
Viral keratitis usually does not present as a “corneal ulcer” (but can, especially with a secondary bacterial or fungal infection) and, as such, is beyond the scope of this article.
When faced with a corneal ulcer, the key to a successful outcome starts with early diagnosis followed by treatment with multiple broad-spectrum therapies in many cases and close follow up.
When diagnosis and treatment are challenging, a cooperative effort with a cornea specialist is the best method of care.
(1).jpg)