The Benefits and Risks of Low Level Light Therapy (Photobiomodulation) in Dry Age-Related Macular Degeneration
Age-related macular degeneration (AMD) is a common condition of the eyes. It usually develops in people aged over 50, and leads to progressive loss of central vision. People with AMD may find it difficult to read or recognise faces, and they can become partially sighted.
Stages of AMD
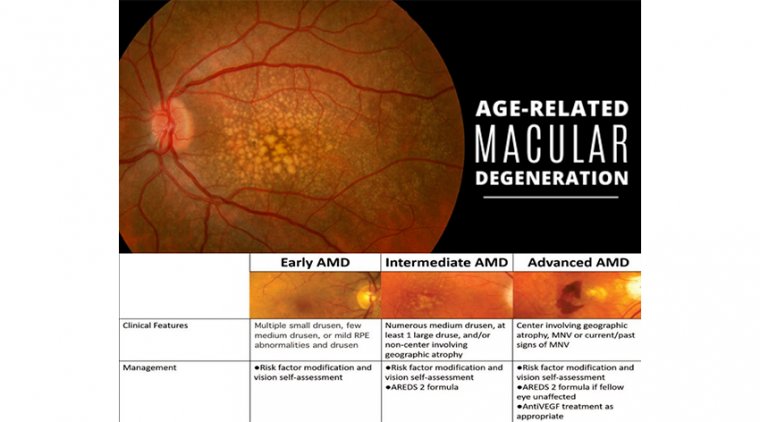
AMD progresses in stages. To begin with, yellow spots (drusen) develop under the retina (the back of the eye). These are not visible to the naked eye, but can be seen by health professionals during examinations of the eyes.
As AMD progresses, cells located in the macula (the central area of the retina) that are needed for vision die. If blood vessels in the eye go on to leak, the condition is classed as ‘wet’ AMD. If there is no leakage, the condition is known as ‘dry’ AMD.
What is Photobiomodulation (PBM)?
Photobiomodulation (PBM) involves the use of visible to near‐infrared (NIR) light (500–1000 nm) produced by a laser or non‐coherent light sources such as light emitting diodes (LEDs) applied to the body to produce beneficial cellular effects.
Light in this range penetrates tissue depending on the wavelength and stimulates cellular function via activation of photoacceptors.
Age‐related macular degeneration (AMD) is a retinal degenerative disease that causes irreversible and profound vision loss in people over the age of 60 years with estimates of AMD patients nearing 50 million worldwide. It is emerging as one of the primary causes of vision impairment in the developed world.
Age‐related macular degeneration (AMD) occurs in two major forms: exudative (wet) and atrophic (dry) AMD. Dry AMD is characterized by drusen, retinal pigment epithelial (RPE) cell atrophy and subjacent photoreceptor degeneration.
Factors involved in causing RPE cell injury and dysfunction have been shown to include mitochondrial dysfunction, oxidative stress, inflammation and genetic disposition.
The vast majority of AMD patients suffering from dry AMD, marked by RPE dysfunction with drusen formation and eventual retinal atrophy, have no effective treatment options other than lifestyle modification and the use of vitamins.
A safe and globally expanding medical intervention is the use of low‐level laser (light) therapy (LLLT) that now includes diabetic wound repair, arthritis, cancer radiation protection (oral mucositis), dental, sports medicine and skeletal muscle disorders (trauma and pain).
Low‐level laser therapy exerts its beneficial effects through increased blood flow and stimulation of cellular functions, a process called photobiomodulation (PBM).
Photobiomodulation (PBM) is the application of monochromatic light to the body with the aim of repairing tissues and reducing inflammation, oedema and pain.
It has been used for 20 years for the treatment of musculoskeletal pain, injury and dysfunction; to aid wound healing; to improve acute muscle performance and reduce muscle damage after exercise; and for neuropathic pain, lymphoedema and oral mucositis.
Several studies in the past 5 years have shown encouraging results using PBM to treat eye diseases including age-related macular degeneration (AMD), retinopathy of prematurity and diabetic macular oedema.
PBM does not worsen the disease, has no side effects and is completely non-invasive. There is no currently approved treatment or cure for the dry form of AMD, which affects 80% of individuals with AMD and tends to progress more slowly than the wet type.
Light waves PBM is not a heat therapy but is more akin to photosynthesis in plants, in that light, in the far red and near-infrared spectral range, can stimulate cells and lead to a cascade of photochemical reactions.
What happens first is that the low-powered light is absorbed locally by cytochrome c oxidase. Mitochondrial energy is then produced by releasing oxygen, which results in increased ATP concentration and reduced oxidative stress.
This photochemical reaction activates enzymes and second messengers, which lead to a cellular and, indirectly, a systemic response in tissues that have not absorbed photons. Mitochondrial dysfunction and oxidative stress play a key role in many macular diseases, so PBM is of use in acute and chronic eye diseases.
We offer nine PBM therapy cycles within approximately 1 month. During the procedure, a medical device with light-emitting diodes stimulate cellular function and improve energy production. Each cycle of therapy delivers wavelengths in the range of 590–850 nm for 4 minutes per eye.
Clinical outcomes are determined immediately after the final cycle, after 3 months and after 6 months, using optical coherence tomography (OCT) imaging, the Amsler grid, the Pelli-Robson chart, a Snellen chart and a Jaeger chart.
Case study Nine PBM cycles were administered over 1 month to a patient with dry AMD, after which OCT showed reduced drusen and the patient obtained subjectively improved vision. The patient also experienced less eye strain, more colour contrast, higher definition and better far and near uncorrected visual acuity.
Contrast sensitivity improved from 1.8 to 2.0 log units. Outcomes remained stable at the 6-month follow-up. This case demonstrated a successful non-invasive treatment with improved quality of vision in dry AMD.
Irradiation could therefore offer a new noninvasive, adverse-effect-free means of stimulating retinal stem cells to regenerate.
To date, there are no approved treatments for many retinal diseases. With its ability to promote cellular regeneration using light waves, PBM has resulted in better visual acuity, contrast sensitivity and a lessdamaged macular profile in several patients with dry AMD.
Overall, these results are encouraging. Our protocol seems to offer an extremely promising approach to prevent visual acuity from worsening and to promote tissue repair in dry AMD. Moreover, this method has the advantage of being entirely non-invasive.
(1).jpg)