Researchers at the University of Rochester have discovered a new benefit of blinking—it enhances vis...
read moreCanadian researchers have evaluated the capabilities of two artificial intelligence chatbots, Google...
read moreIn a study published in the Journal of Neuroscience, researchers at the University of Rochester have...
read moreResearchers at the National Institutes of Health have significantly advanced retinal imaging by inte...
read moreLenz Therapeutics announced positive topline results from its Phase 3 CLARITY study, evaluating two ...
read moreResearchers discovered that individuals vary significantly in their ability to perceive visual signa...
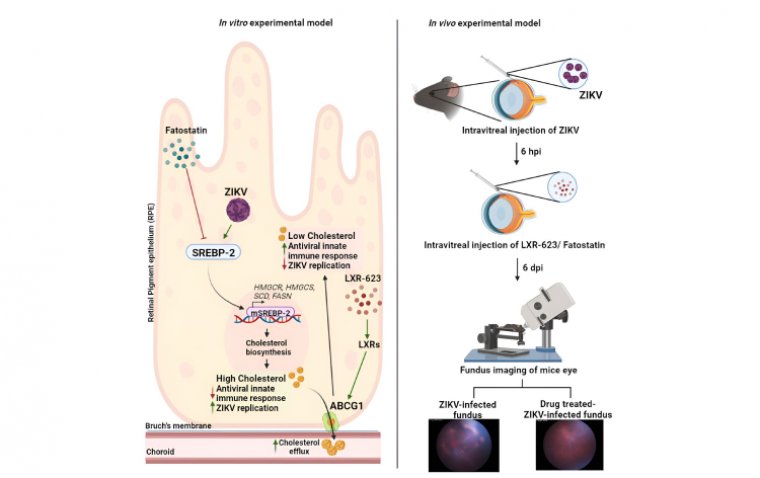
read moreResearchers at Wayne State University School of Medicine have published a study in the journal iScie...
read moreRegenxbio Inc. announced the publication of their phase 1/2a study results in The Lancet, showcasing...
read moreAurion Biotech recently administered its novel cell therapy, AURN001, to the first Canadian particip...
read moreNanoscope Therapeutics announced positive topline results from the phase 2b RESTORE trial, evaluatin...
read more More
More